Mal de Débarquement Syndrome (MdDS)
What is Mal de Débarquement Syndrome (MdDS)?
“Mal de Débarquement Syndrome is characterized by a persistent sensation of rocking, swaying, or bobbing that occurs when an individual is stationary” (Saha & Fife, 2015).
Symptoms commonly begin after exposure to prolonged motion, such as boat travel, flying, or long car rides. Unlike the temporary symptoms that many people experience after travel, symptoms of MdDS persist for weeks, months, or longer (Van Ombergen et al., 2016; Saha & Cha, 2020).
History
-
“Mal de débarquement” is French for “sickness of disembarkation.”
-
Temporary rocking sensations after sea travel have been described for centuries among sailors and are often called “sea legs.”
-
In most people, these sensations resolve within hours to a few days as the brain readapts to stable ground.
-
Persistent symptoms were later recognized as a distinct clinical disorder when the sensation does not resolve after travel.
-
Diagnostic criteria were proposed in 2015, helping standardize recognition of MdDS (Saha & Fife, 2015).

Etiology
The exact cause of MdDS is not fully understood. Current evidence suggests it may represent a disorder of neuroplasticity and vestibular adaptation, in which the brain has difficulty switching out of an adapted motion state after travel has ended (Saha & Fife, 2015; Van Ombergen et al., 2016).
🧠 Vestibular adaptation that does not reset – The brain normally adapts to motion during travel. In MdDS, this adaptation may persist after the motion stops (Saha & Fife, 2015).
🔄 Changes in brain connectivity – Neuroimaging studies suggest altered activity and connectivity in brain networks involved in motion perception (Cha et al., 2021; Saha & Cha, 2020).
Prevalence
The true prevalence of Mal de Débarquement is not well established. The condition is considered rare and likely underdiagnosed, partly because symptoms can be mistaken for other vestibular or neurologic disorders (Van Ombergen et al., 2016; Saha & Cha, 2020).
Patterns have been identified in research:
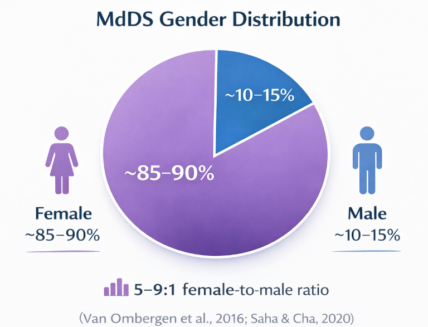
- Women are affected far more often than men, particularly in middle age. Studies report that approximately 85–90% of individuals with MdDS are female, compared with 10–15% male (Van Ombergen et al., 2016; Saha & Cha, 2020).
- Many reported cases occur between ages 30–60, though MdDS can occur at other ages (Van Ombergen et al., 2016).
Patient Reported Symptoms
Patients with MdDS commonly report the following symptoms:
🚢 Rocking, swaying, or bobbing sensation, often described as feeling like being on a boat (Saha & Fife, 2015; Van Ombergen et al., 2016)
🌀 Persistent perception of motion while sitting, standing, or lying still (Saha & Cha, 2020)
🚗 Symptoms that improve during passive motion, such as riding in a car (Saha & Fife, 2015; Van Ombergen et al., 2016)
⚖️ Balance difficulties or unsteadiness (Van Ombergen et al., 2016)
🧠 Cognitive symptoms, including difficulty concentrating or “brain fog” (Van Ombergen et al., 2016; Saha & Cha, 2020)
😴 Fatigue and reduced tolerance for visually busy environments (Van Ombergen et al., 2016)
🤕 Headache or migraine symptoms in some individuals (Saha & Cha, 2020; Beh, 2022)
📅 Symptoms may persist for months or years, significantly impacting daily activities and quality of life (Van Ombergen et al., 2016).
Clinical Findings
Clinical evaluation often reveals few objective abnormalities on routine neurologic or vestibular examination. Diagnosis is therefore based primarily on the characteristic clinical history and symptom pattern rather than a definitive diagnostic test (Saha & Fife, 2015; Saha & Cha, 2020; Van Ombergen et al., 2016).
Typical examination findings may include:
-
Neurologic examination is usually benign without focal neurologic deficits (Saha & Fife, 2015).
-
Videonystagmography (VNG) typically demonstrates normal vestibular responses (Saha & Fife, 2015).
-
Hearing is generally normal on audiologic testing (Saha & Fife, 2015).
-
Structural brain imaging is typically normal and does not identify a specific structural abnormality responsible for symptoms (Saha & Fife, 2015).
-
Some patients may appear hesitant during balance testing, despite limited objective abnormalities (Saha & Fife, 2015).
Neuroimaging Research Findings
Research using advanced functional brain imaging has identified changes in brain activity and connectivity in individuals with persistent MdDS.
Key findings include:
-
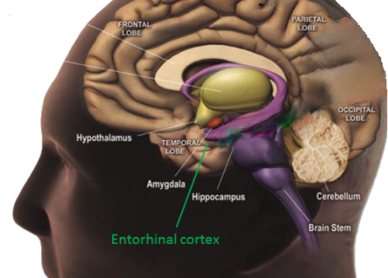
Increased metabolic activity in the left entorhinal cortex and amygdala, brain regions involved in spatial navigation and memory processing (Cha, Ding, & Yuan, 2021).
-
Altered functional connectivity between these regions and other sensory-processing areas of the brain, including increased connectivity with posterior sensory regions and reduced connectivity with frontal and temporal cortices (Cha, Ding, & Yuan, 2021).
These findings suggest that MdDS may involve maladaptation of central brain networks involved in motion perception and spatial orientation, rather than a primary peripheral vestibular disorder (Saha & Cha, 2020; Cha, Ding, & Yuan, 2021).
Vestibular Findings
MdDS usually does not show clear vestibular test abnormalities.
That is why it is frequently misdiagnosed as:
-
anxiety disorders
This point is strongly emphasized in the review literature (Van Ombergen et al., 2016).
Treatment
Treatment focuses on improving how the brain processes motion. No single treatment is effective for all patients, and several approaches remain under investigation.
1. Optokinetic / Visual Motion Therapy
-
Uses moving visual patterns combined with head movements
-
Designed to readapt abnormal motion perception
-
A standardized approach using head roll (side to side head motion) while viewing moving visual lines has been reported to improve symptoms in approximately two-thirds of patients (Schoenmaekers et al., 2024; Hoppes et al., 2021)
-
Sham-controlled research also supports improvement with optokinetic stimulation compared to control conditions (Mucci et al., 2018)
2. Neuromodulation (Emerging Treatments)
-
Includes:
-
Repetitive transcranial magnetic stimulation (TMS)
-
Transcranial alternating current stimulation (tACS)
-
Galvanic vestibular stimulation (GVS)
-
-
These approaches target brain regions involved in motion perception and spatial processing
-
Studies demonstrate potential symptom improvement, but these treatments remain investigational and require further research (Cha et al., 2022; Schoenmaekers et al., 2025)
3. Vestibular Rehabilitation
-
Outcomes are variable, and no single standardized approach has been established (Kinne et al., 2022)
-
Evidence suggests:
-
Traditional gaze stabilization (VOR x1) is not effective for MdDS
-
Treatment should focus on:
-
Increasing reliance on proprioceptive input
-
-
These strategies are supported by case-based evidence (Liphart, 2015)
4. Medical Management
-
There is no single standardized medication treatment for MdDS
-
Some medications have been reported to help in certain patients, including:
-
Venlafaxine
-
Anti-epileptic medications
-
Vitamin supplementation (e.g., B2)
-
-
These are based on clinical observations and related vestibular literature (Beh et al., 2021)
Clinical Considerations
- Motion-triggered MdDS has been reported to respond more favorably to treatment than spontaneous-onset MdDS, based on observational and review data (Van Ombergen et al., 2016; Saha and Cha, 2020)
-
Symptoms often improve temporarily during passive motion (e.g., driving), which is a characteristic clinical feature used in diagnosis (Saha and Fife, 2015; Saha and Cha, 2020)
-
Patients with MdDS often have normal or non-specific findings on standard vestibular testing, supporting the concept that this is not a typical peripheral vestibular disorder (Van Ombergen et al., 2016; Cha et al., 2021)
-
MdDS is thought to involve persistent maladaptive vestibular adaptation and altered sensory integration, which is why treatments focus on recalibration rather than compensation (Cha et al., 2021; Saha and Fife, 2015)
-
Treatment response is variable, and the overall evidence base remains limited, with few randomized controlled trials and small sample sizes (Kinkhabwala et al., 2023)
To Learn More – Check out these Resources
- Vestibular First’s Online, self-paced CEU course: Vestibular Examination: For Clinical Practice
- Bundle and SAVE on all courses!
- Watch related Journal Clubs:
- Printable resources for patients:
- Learn more:
References
- Beh, S.C. Vestibular Migraine. Curr Neurol Neurosci Rep 22, 601–609 (2022). https://doi.org/10.1007/s11910-022-01222-6
- Cha YH, Deblieck C, Wu AD. Double-Blind Sham-Controlled Crossover Trial of Repetitive Transcranial Magnetic Stimulation for Mal de Debarquement Syndrome. Otol Neurotol. 2016 Jul;37(6):805-12. doi: 10.1097/MAO.0000000000001045. PMID: 27176615; PMCID: PMC4907861. https://pubmed.ncbi.nlm.nih.gov/27176615/
- Cha YH, Ding L, Yuan H. Neuroimaging Markers of Mal de Débarquement Syndrome. Front Neurol. 2021 Mar 4;12:636224. doi: 10.3389/fneur.2021.636224. PMID: 33746890; PMCID: PMC7970001. https://pubmed.ncbi.nlm.nih.gov/33746890/
- Cha YH, Gleghorn D, Doudican BC. Double-blind randomized N-of-1 trial of transcranial alternating current stimulation for mal de débarquement syndrome. PLoS One. 2022 Feb 4;17(2):e0263558. doi: 10.1371/journal.pone.0263558. Erratum in: PLoS One. 2024 Dec 10;19(12):e0315783. doi: 10.1371/journal.pone.0315783. PMID: 35120184; PMCID: PMC8815977. https://pubmed.ncbi.nlm.nih.gov/35120184/
- Hoppes CW, Vernon M, Morrell RL, Whitney SL. Treatment of Mal de Debarquement Syndrome in a Computer-Assisted Rehabilitation Environment. Mil Med. 2022 Jul 1;187(7-8):e1011-e1015. doi: 10.1093/milmed/usab077. PMID: 33604663. https://vestibularfirst.com/wp-content/uploads/2021/10/Mal-de-Debarquement-Hoppes-2021.pdf?srsltid=AfmBOoo9hLIA-FYYdfuPKiUB2ghyNPoxs2Sn4WsBHW7Xfr2gpEHJ4wTN
- Kinne, B. L., Bode, E. R., Laisure, S. S., & Schmitt, J. R. (2022). Vestibular rehabilitation for individuals with mal de debarquement syndrome: a systematic review. Physical Therapy Reviews, 27(3), 205–213. https://doi.org/10.1080/10833196.2022.2026007
- Liphart J. Use of sensory reweighting for a woman with persistent mal de debarquement: a case report. J Geriatr Phys Ther. 2015 Apr-Jun;38(2):96-103. doi: 10.1519/JPT.0000000000000022. PMID: 24978933. https://pubmed.ncbi.nlm.nih.gov/24978933/
- Mucci V, Perkisas T, Jillings SD, Van Rompaey V, Van Ombergen A, Fransen E, Vereeck L, Wuyts FL, Van de Heyning PH, Browne CJ. Sham-Controlled Study of Optokinetic Stimuli as Treatment for Mal de Debarquement Syndrome. Front Neurol. 2018 Oct 25;9:887. doi: 10.3389/fneur.2018.00887. PMID: 30410464; PMCID: PMC6210740. https://pubmed.ncbi.nlm.nih.gov/30410464/
- Saha KC, Fife TD. Mal de débarquement syndrome: Review and proposed diagnostic criteria. Neurol Clin Pract. 2015 Jun;5(3):209-215. doi: 10.1212/CPJ.0000000000000116. PMID: 29443215; PMCID: PMC5764463. https://pmc.ncbi.nlm.nih.gov/articles/PMC5764463/
- Saha K, Cha YH. Mal de Debarquement Syndrome. Semin Neurol. 2020 Feb;40(1):160-164. doi: 10.1055/s-0039-3402740. Epub 2020 Jan 27. PMID: 31986543. https://pubmed.ncbi.nlm.nih.gov/31986543/
- Schoenmaekers C, Jillings S, De Laet C, Zarowski A, Wuyts FL. Guideline for standardized approach in the treatment of the Mal de Debarquement syndrome. Front Neurol. 2024 Mar 19;15:1359116. doi: 10.3389/fneur.2024.1359116. PMID: 38566854; PMCID: PMC10985174. https://pubmed.ncbi.nlm.nih.gov/38566854/
- Schoenmaekers C, De Smet D, Peeters S, Zarowski A, Deblieck C, Wuyts FL. Galvanic vestibular stimulation for Mal de Debarquement syndrome: a pilot study on therapeutic potential. Exp Brain Res. 2025 May 14;243(6):145. doi: 10.1007/s00221-025-07102-y. PMID: 40369206; PMCID: PMC12078377. https://pubmed.ncbi.nlm.nih.gov/40369206/
- Van Ombergen A, Van Rompaey V, Maes LK, Van de Heyning PH, Wuyts FL. Mal de debarquement syndrome: a systematic review. J Neurol. 2016 May;263(5):843-854. doi: 10.1007/s00415-015-7962-6. Epub 2015 Nov 11. PMID: 26559820; PMCID: PMC4859840. https://pubmed.ncbi.nlm.nih.gov/26559820/